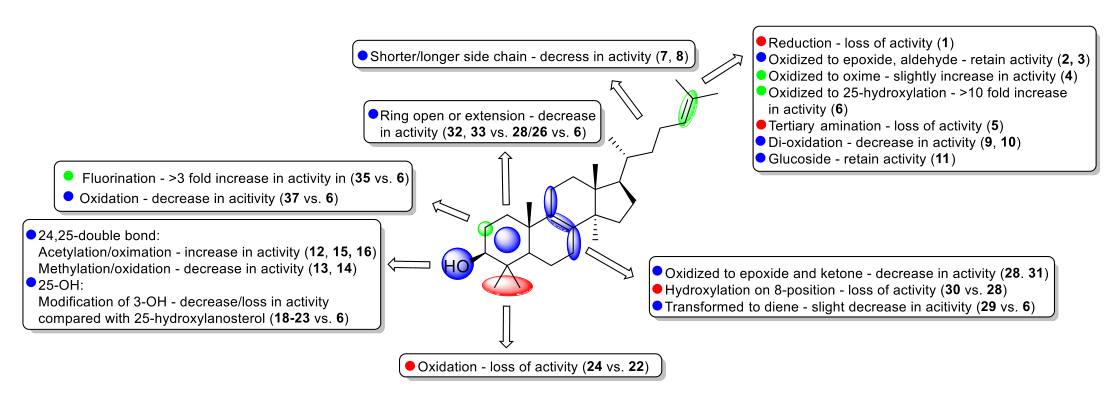
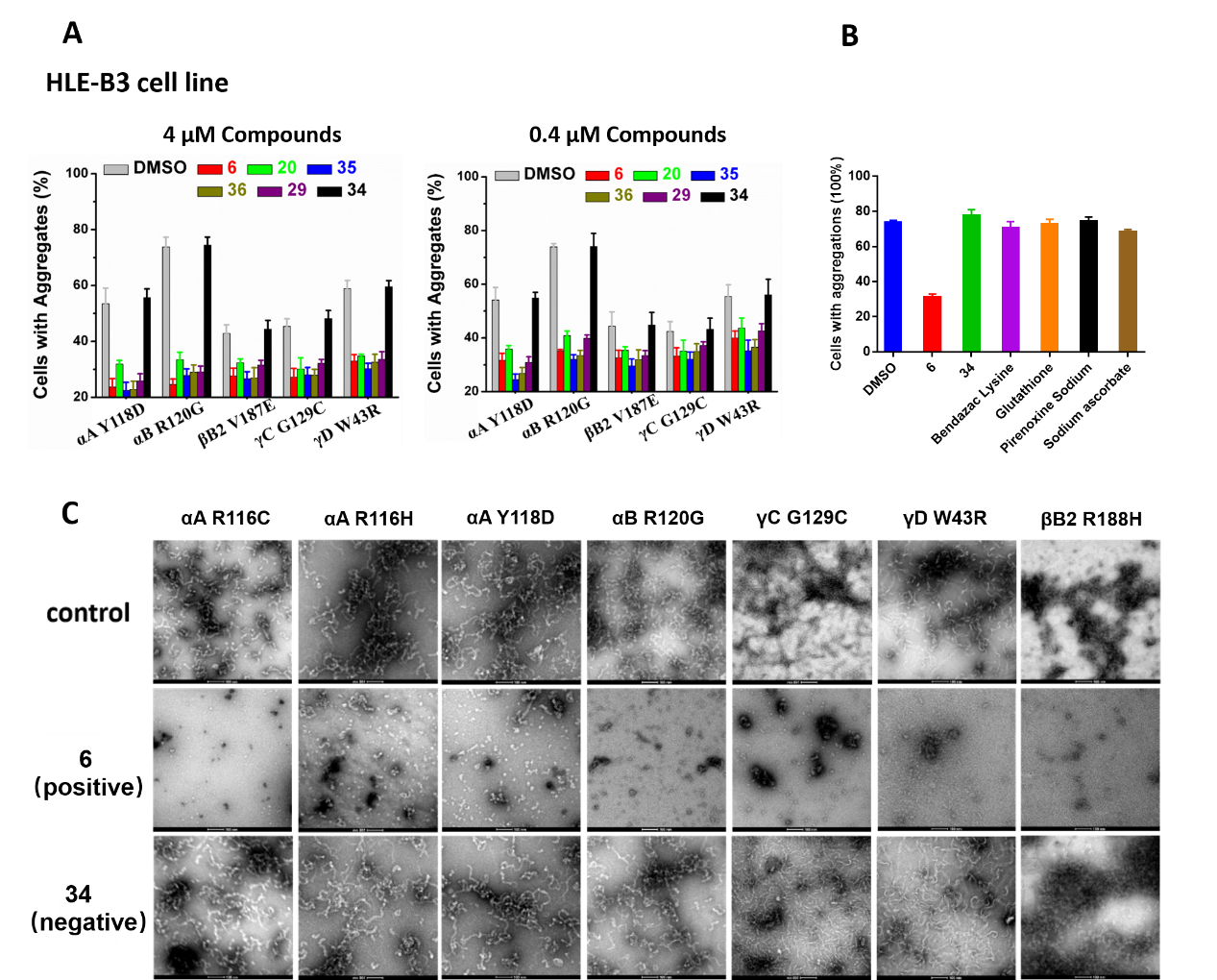
We describe here the development of potent synthetic analogues of naturally occurring triterpenoid lanosterol to reverse protein aggregation in cataracts. Lanosterol showed superiority to other scaffolds in terms of efficacy and generality in previous studies. Various modified lanosterol derivatives were synthesized via modification of the side chain, ring A, ring B and ring C. Evaluation of these synthetic analogues draws a clear picture for SAR. In particular, hydroxylation of the 25-position in the side chain profoundly improved the potency, and 2-fluorination further enhanced the biological activity. This work also revealed that synthetic lanosterol analogues could reverse multiple types of mutant crystalline aggregates in cell models with excellent potency and efficacy. Notably, lanosterol analogues have no cytotoxicity but can improve the viability of the HLE-B3 cell line. Furthermore, representative compound 6 successfully redissolved the aggregated crystallin proteins from the amyloid-like fibrils in a concentration-dependent manner.